-
Courses

Courses
Choosing a course is one of the most important decisions you'll ever make! View our courses and see what our students and lecturers have to say about the courses you are interested in at the links below.
-
University Life

University Life
Each year more than 4,000 choose University of Galway as their University of choice. Find out what life at University of Galway is all about here.
-
About University of Galway

About University of Galway
Since 1845, University of Galway has been sharing the highest quality teaching and research with Ireland and the world. Find out what makes our University so special – from our distinguished history to the latest news and campus developments.
-
Colleges & Schools

Colleges & Schools
University of Galway has earned international recognition as a research-led university with a commitment to top quality teaching across a range of key areas of expertise.
-
Research & Innovation

Research & Innovation
University of Galway’s vibrant research community take on some of the most pressing challenges of our times.
-
Business & Industry

Guiding Breakthrough Research at University of Galway
We explore and facilitate commercial opportunities for the research community at University of Galway, as well as facilitating industry partnership.
-
Alumni & Friends

Alumni & Friends
There are 128,000 University of Galway alumni worldwide. Stay connected to your alumni community! Join our social networks and update your details online.
-
Community Engagement

Community Engagement
At University of Galway, we believe that the best learning takes place when you apply what you learn in a real world context. That's why many of our courses include work placements or community projects.
Light Microscopy
Overview
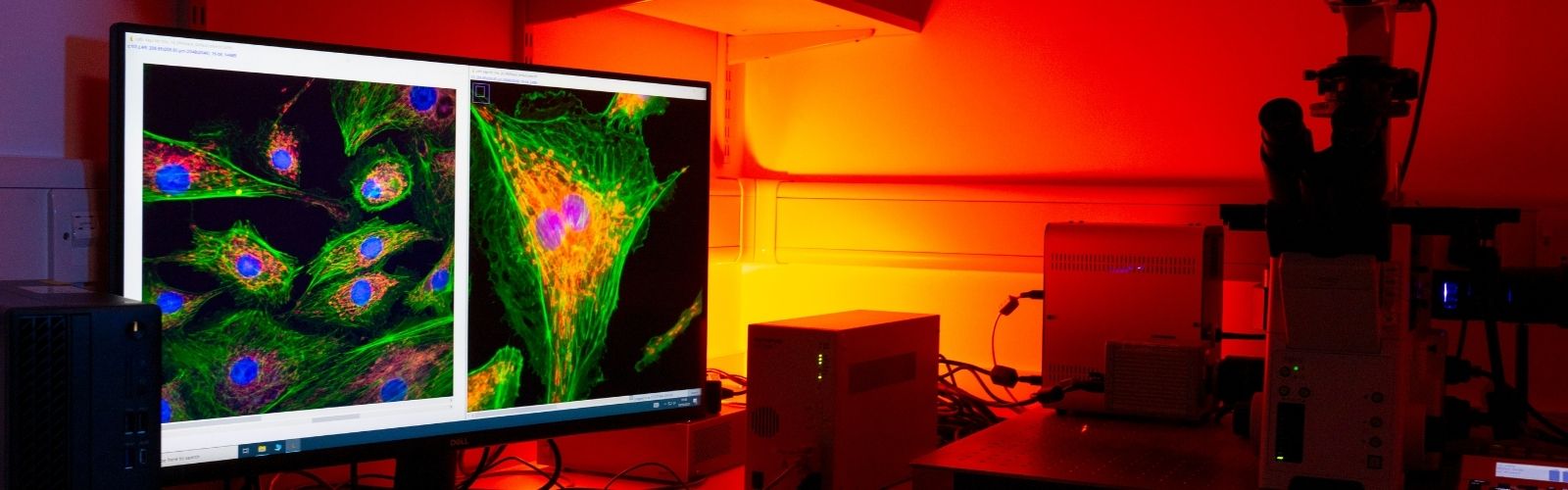
The Light Microscopy Facility specialises in basic and advanced fluorescence microscopy techniques. Microscopy is the gold standard technique for the morphological analysis of cells and tissues. We provide researchers with access to advanced imaging instruments, technical expertise, and training. Its purpose is to enable high-quality, reproducible imaging for a wide range of biological and materials science applications. Refer to the equipment summary dropdown to discover the capabilities of our microscopes.
The facility provides:
- Advice on selecting the right microscope and imaging modalities
- In-depth training for autonomous instrument operation
- Guidance on experimental design, sample preparation, image acquisition, and optimization
- Assistance with complex or cutting-edge imaging techniques
- Help with troubleshooting and data interpretation
- Workstations and software for quantitative image analysis (e.g., FIJI/ImageJ, Huygens)
- Support with segmentation, tracking, colocalization, 3D reconstruction, machine-learning–based analysis
- Data storage solutions and workflow guidance for large imaging datasets
Our overall mission is to enable high-quality, cutting-edge imaging by:
- Making expensive instruments accessible to many researchers
- Ensuring proper training and reproducibility
- Providing expert guidance to maximize scientific impact
Light Microscopy Core Facility Equipment
Equipment Summary
Table 1. Microscope types in the Core Facility.
| Nikon Eclipse E600 | Olympus IX81 | Olympus FV1000 | Olympus FV3000 | Leica Stellaris Dive Falcon | Zeiss Elyra7 Lattice SIM | |
| Inverted/Upright | Upright | Inverted | Inverted | Inverted | Upright | Inverted |
| Widefield Epifluorescence | Yes | Yes | - | - | - | Yes |
| Brightfield | Yes | Yes | Yes | Yes | Yes | Yes |
| Darkfield | Yes | - | - | - | - | - |
| Differential Interference Contrast (DIC) | - | - | - | Yes | - | Yes |
| Laser Scanning Confocal | - | - | Yes | Yes | Yes | - |
| Multiphoton | - | - | - | - | Yes | - |
| Fluorescence Lifetime Imaging (FLIM) | - | - | - | - | Yes | - |
| Structured Illumination Microscopy (SIM) | - | - | - | - | - | Yes |
| Super Resolution | - | - | - | - | - | Yes |
Table 2. Experiment / Sample types each microscope can work with. *System is upright, not a typical live cell setup.
| Nikon Eclipse E600 | Olympus IX81 | Olympus FV1000 | Olympus FV3000 | Leica Stellaris Dive Falcon | Zeiss Elyra7 Lattice SIM | |
| Timelapse imaging with temperature, O2 and CO2 control | - | - | - | Yes | Yes* | Yes |
| Fast timelapse imaging | - | - | - | - | - | Yes |
| Fixed/unfixed cells/spheroids in multi-well dishes | - | Yes | Yes | Yes | - | Yes |
| Fixed cells on slides | Yes | Yes | Yes | Yes | Yes | Yes |
| Small spheroids/organoids | - | - | Yes | Yes | Yes | - |
| Large spheroids/organoids | - | - | - | - | Yes | - |
| Thin tissue sections | - | - | Yes | Yes | Yes | Yes |
| Thick tissue sections | - | - | Yes | Yes | Yes | - |
| Large gels with cells/spheroids or cleared tissue | - | - | - | - | Yes | - |
For a more comprehensive breakdown of each microscope, click on the equipment names below.
Zeiss Elyra 7 Lattice SIM
Type / technique: Inverted super-resolution structured illumination microscope
The Zeiss Elyra 7 microscope achieves super resolution from the structured Illumination process along with computer processing and deconvolution. This is a fast, camera-based system, and can be used for both fixed and live samples, and is compatible with multiwell dishes.
Super resolution imaging is achieved using the 63x oil lens. Here a lattice blocks out some of the illumination light, the lattice is rotated and multiple images of the same field of view are acquired. The images acquired when the lattice was in different phases are used to reconstruct a super-resolution image. The SIM2 algorithm may also be applied to further increase resolution using deconvolution. Outside of super resolution the system can also be used to generate confocal-like images with optical sectioning using the Apotome mode. Here a striped grid blocks out some of the illumination light, like above, images are acquired with the grid in different phases. These images are then processed to better resolve structures of interest.
- Microscope base: Axio Observer 7 inverted
- Lightsource: 405, 488, 561 and 642 nm lasers
- Detection: 2 Hamamatsu ORCA-fusion BT cameras
- Objective lenses: 2.5X, 10X, 25X (multi-immersion), 40X (oil), 40X (water), 63X (oil), 63X (water)
- Transmitted light: brightfield, DIC (using 10X or 63X water)
- XYZ control: Motorized XYZ control
- Incubation temp and CO2 control: yes
- Optical sectionins: yes, structured illumination lattice or apotome mode
- Software: ZenBlue
Leica Stellaris Dive Falcon
Type / technique: Confocal, multiphoton, fluorescence lifetime imaging.
The Leica Stellaris Dive is a microscope with dual functionality. It is in an upright configuration and can be used for regular laser scanning confocal microscopy as well as multiphoton microscopy. The change from confocal to multiphoton mode is seamless and completed by the STO, it consists of activating the multiphoton lasers and changing the nosepiece of the microscope so that it houses the 25X multiphoton water dipping lens. As a confocal microscope, due to its upright configuration it is most suitable for fixed samples which are mounted with a coverslip rather than samples in multiwell dishes. If the samples are three dimensional, they can be mounted with silicone spacers and imaged using confocal or multiphoton mode.
Where the system really shines is in multiphoton mode. Samples like spheroids encased in gels are imaged in petri dishes in an aqueous solution such as PBS, the multiphoton lens dips directly into this solution. The system is designed for deep cell/tissue imaging, the multiphoton lens has a long working distance and the theoretical z limit for imaging is in the mm range, although the actual results will depend on the density of the sample and depth of penetration of the fluorophores. The system in multiphoton mode can also be used to image thick mounted and cleared samples of tissue by simply placing a large drop of water on-top of the coverslip. The system is also equipped with FAst Lifetime CONtrast (FALCON) module for FLIM experiments. FLIM is a technique which can be used to separate fluorescence emissions which overlap in the same channel, for example tissue autofluorescence and GFP expression. This is due to differences in the fluorescence lifetime properties molecules which auto fluoresce and dyes of interest.
- Microscope base: Leica DM6 upright
- XYZ control: Motorized XYZ control
- Incubation temp and CO2 control: yes
- Software: LAS-X
Laser Scanning Confocal
- Lightsource: 405, 488, 561 and 638 nm lasers
- Detection: Tuneable emission using HyD S 1, HyD S 2, HyD S 3, HyD X 4 detectors
- Scanner: Galvo (standard) or resonant (fast)
- Objective lenses: 10x, 20x (multi-immersion), 40x (glycerol), 63x (oil).
Multiphoton
- Lightsource: tuneable 680-1300nm and a 1045nm femtosecond pulsed lasers (Insight dual IR)
- Objective lens: 25x long working distance water dipping lens with motorised correction collar
- Detection: Tuneable emission using 3 non-descanned detectors located withing Deep In-Vivo Explorer (DIVE) module
Olympus FV3000
Type: Inverted laser scanning confocal microscope
The Olympus FV3000 is an inverted laser scanning confocal microscope which is perfect for imaging both live or fixed biological samples. The system is equipped with DIC and allows for simultaneous imaging using DIC and fluorescence channels. It is equipped with an incubation chamber and CO2/O2 control and so is suitable for time-lapse imaging. The system also has a resonant scanner which can be used for fast live cell imaging. The emission is detected using 4 detectors allowing for simultaneous imaging of four spectrally separated channels, while the detectors themselves can be tuned to specific emission ranges. This system was installed back in 2018 and quickly became the workhorse of the facility due to its to its user-friendly design as well as versatility in the types of samples that it can image. This system is well suited for imaging samples in multi-well dishes, fixed slides of cells or tissues and even small spheroids/organoids.
- Microscope base: Inverted Olympus IX81
- Lightsource: 405, 488, 543 and 633 nm lasers
- Detection: 2 high sensitivity (HSD) and 2 standard PMT detectors (SD)
- Scanner: Resonant (high speed) and Galvo (standard)
- Objective lenses: 2x, 4x, 10x, 20x (air or oil), 30x (silicone), 40x (air, water or silicone), 60x (oil), 100x (oil)
- Transmitted light: brightfield, DIC (compatible with LSM)
- Emission: Tuneable detection range down to 1nm accuracy
- XYZ control: Motorized XYZ control
- Incubation temp and CO2 control: yes
- Software: Fluoview
Witec Alpha500
Type / technique: Raman Spectroscopy
This system is used to generate raman spectra from a wide variety of materials from inorganic to complex biomaterials and tissues. The system can scan a fixed point, 2 d maps and also scan in z. Very little sample preparation is involved, quartz is the recommended substrate for holding the samples of interest. We primarily run assisted sessions on this instrument but is available for autonomous use pending extensive training.
- Microscope Base: upright frame gantry arrangement
- Lightsource: XTRA high power single frequency diode 785nm laser
- Optical sectioning: yes confocal
- Objective lenses: 10x, 20x, 40x, 60x, 100X
- XYZ control: Motorized XYZ control
Olympus IX81
Type: Inverted Widefield Fluorescence System
The Olympus IX81 is an inverted fluorescence microscope which uses a 16 colour LED light source and traditional DAPI, FITC, TRITC and Cy5 filter sets. The system is equipped with motorized z control allowing users to acquire 3D z-stacks in multiple channels. In 2022 the system the system was fitted with a modern LED light source, new camera, new PC, and opensource software MicroManager for acquisition of images.
- Microscope base: Inverted Olympus IX81
- Lightsource: COOLLED pE-4000, 16 colour LED source
- Detection: Hamamatsu ORCA-Flash4.0L C11440 digital camera
- Objective lenses: 4X, 10X, 20X, 40X (oil), 60X (oil) 100X (oil)
- Epi fluorescence: DAPI, FITC, TRITC, Cy5 classic filter sets
- XYZ control: Manual XY, motorized Z control
- Software: MicroManager (open source)
Olympus FV1000
Type: Inverted laser scanning confocal microscope
The Olympus FV1000 is an inverted laser scanning confocal microscope which is perfect for imaging both live or fixed biological samples. The system is not equipped with an incubation chamber and so is not suitable for time-lapse imaging which requires incubation and CO2/O2 control. Although it is an older system it still functions well and is the only laser scanning confocal system located in the biomedical sciences building.
- Microscope base: Inverted Olympus IX71
- Lightsource: 405, 473, 546, 633nm excitation lasers
- Detection: 3 tuneable PMT detectors
- XYZ control: Motorized XYZ control
- Software: Evident Fluoview
Nikon Eclipse
Type: Upright Widefield Fluorescence System with a Darkfield Setup
The Nikon Eclipse E600 is an upright fluorescence microscope which uses an LED light source and classical DAPI, FITC, TRITC filter sets. In 2016 the system was upgraded with a dark-field set-up while other optics and camera was upgraded in 2024.
- Microscope base: Upright Nikon Eclipse 5600
- Lightsource: PrecisExcite COOLLED light source
- Detection: TUCSEN FL20BW colour camera
- Transmitted light: darkfield condenser and 100x darkfield optimised lens
- Epifluorescence: DAPI, FITC, TRITC classic filter sets
- XYZ control: All manual
- Software: Cell Sens
For more information, please contact microscopy@universityofgalway.ie