-
Courses

Courses
Choosing a course is one of the most important decisions you'll ever make! View our courses and see what our students and lecturers have to say about the courses you are interested in at the links below.
-
University Life

University Life
Each year more than 4,000 choose University of Galway as their University of choice. Find out what life at University of Galway is all about here.
-
About University of Galway

About University of Galway
Since 1845, University of Galway has been sharing the highest quality teaching and research with Ireland and the world. Find out what makes our University so special – from our distinguished history to the latest news and campus developments.
-
Colleges & Schools

Colleges & Schools
University of Galway has earned international recognition as a research-led university with a commitment to top quality teaching across a range of key areas of expertise.
-
Research & Innovation

Research & Innovation
University of Galway’s vibrant research community take on some of the most pressing challenges of our times.
-
Business & Industry

Guiding Breakthrough Research at University of Galway
We explore and facilitate commercial opportunities for the research community at University of Galway, as well as facilitating industry partnership.
-
Alumni & Friends

Alumni & Friends
There are 128,000 University of Galway alumni worldwide. Stay connected to your alumni community! Join our social networks and update your details online.
-
Community Engagement

Community Engagement
At University of Galway, we believe that the best learning takes place when you apply what you learn in a real world context. That's why many of our courses include work placements or community projects.
Research Interests
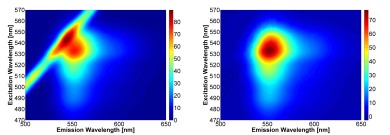 The research in the laboratory focuses on two core areas: Process Analytical Technologies/Bio-analytical, and Advanced Microscopy & Fluorescence Spectroscopy.
The research in the laboratory focuses on two core areas: Process Analytical Technologies/Bio-analytical, and Advanced Microscopy & Fluorescence Spectroscopy.
Within each of these core areas there are a number of different projects which span domains like small molecule analytics, industrial process monitoring, and fluorescence spectroscopy.
PROCESS ANALYTICAL TECHNOLOGIES
The core concepts we use are spectroscopy (interaction of light with matter) and chemometrics (mathematical analysis). The work involves both applied and fundamental research studies with a goal of developing novel, robust, and sensitive analytical methods. We generally focus on the use of Raman and Fluorescence spectroscopies, because they offer an ideal combination of speed, molecular recognition, minimal sample preparation, inexpensive hardware, and robustness. Some of our methods are now being adopted by our industrial partners.
Current research areas include:
-
- Polarized Excitation Emission Matrix (pEEM) Spectroscopy for protein analysis: The pEEM measurement methodology is being developed for the analysis of proteins and protein based assemblies with a focus on applications in biopharmaceutical manufacturing. pEEM measurements provide additional information about size changes in proteins which can be harvested for the analysis of monoclonal antibodies (mAbs), insulin, liposome-protein interactions, and even vaccines.
- Biopharmaceutical Process Analysis: This research area which kicked off in 2006 was focused on the development of novel analytical methods for the rapid, inexpensive, analysis of cell culture media and the raw materials used in industrial biotechnology. Since then it has expanded to collaborations with a variety of multinational companies and covers everything in modern bioprocesses. Current research topics are solid-state media analysis, liquid state media analysis, protein characterization, bioprocess monitoring, and protein-surface interaction measurement. The first project in this area was a collaboration with Bristol Myers-Squibb and DCU, involves the development of new, novel, rapid analytical techniques for the BioPharma industry. The research which involved a variety of spectroscopic techniques (Raman, IR, NIR, XRF, etc) in combination with chemometrics was undertaken as part of the Centre for Bioanalytical Sciences (CBAS). Many of these activities involve collaborations with biopharma companies like Janssen-Biologics, Merck, Bristol Myers Squibb,and instrumentation companies like Kaiser Optical Systems Inc., Horiba, and Agilent. More details of the research are on the bioanalytical page.
- Real-Time-Release (RTR) and low-level polymorph and Impurity analysis of Active Pharmaceutical Ingredients (APIs): This was part of the SFI funded Synthesis and Solid-State Pharmaceutical Centre. A vital issue associated with the manufacture and analysis of solid state APIs and their formulations is the detection and quantification of chemical contaminants and alternate API polymorphs, present at low concentrations. For the RTR of APIs one must validate the quality of the materials in terms of precisely defined contamination limits which is often accomplished by time-consuming methods like High-Performance Liquid Chromatography (HPLC) or other wet chemistry-based methods. Being able to replace these traditional methods with on/in-line spectroscopic based methods will provide significant cost savings to the global PharmaChem sector. The research focuses on the use of Raman spectroscopy and novel chemometric methods. in 2015 we published the first technical method which demonstrated a Limit of Detection (LoD) of 0.03% for a binary powder mixture. The next phase of this research aims to extend the method to lower LoDs and more complex multi-component solid mixtures.
- Low-content quantification in powders using Raman spectroscopy: a facile chemometric approach to sub 0.1% limits of detection., B. Li, A. Calvet, Y. Casamayou-Boucau, C. Morris, and A.G. Ryder, Analytical Chemistry, 87(6), 3419-3428, (2015). DOI: 10.1021/ac504776m
Advanced Microscopy & Fluorescence Spectroscopy
This work involves the development of new instrumentation, applied, and fundamental research. It currently encompasses applications in both the biomedical and earth sciences.
Specific research topics include:
- Anisotropy Resolved Multi-dimensional Emission Spectroscopy (ARMES): This is a novel tool being developed in our lab which seeks to develop a completely new measurement methodology for protein analysis. The research includes aspects of physics (photonics), analytical chemistry, biochemistry, and mathematics (chemometrics).
- Protein-Surface Chemistry: Development of novel fluorescence based methods for the analysis of protein deposition on surfaces. In this project we seek to use the most advanced and sensitive microscopy techniques to study in detail how protein structure and activity changes on different surfaces. The current research involves the use of a Total Internal Reflection Fluorescence (TIRF) microscope.
- Biomedical Polymers: Studying the influence of surface polarity on the luminescence properties of fluorophores with a view to developing analysis tools for the evaluation of biomedical polymers. This also includes looking at protein-polymer interactions in solution and also understaning polymer aggregation.
- Triazine Fluorophore photophysics: This fundamental project is exploring the photophysics of novel 1,2,3, triazine based fluorophores in different environments such as proteins, polymers, and micelles.
- Advanced Fluorescence Microscopy: Fluorescence Correlation Spectroscopy (FCS) and Fluorescence Cross Correlation Spectroscopy (FCCS) for the study of protein interactions with nanoparticles.
Academic Collaborations:
The group collaborated widely with academic researchers in the University of Galway and Internationally. Some of our past collaborators (not an exhaustive list) include:
- Dr. John Stephens, School of Chemistry, NUI Maynooth. We are working together on the photo-physical characterization of novel triazine fluorophores that John synthesizes.
- Dr. Leigh F. Jones, University of Wolverhampton. Spectroscopic analysis of inorganic complexes [see publication list for more details].
- Prof. P. Dockery, Dept. of Anatomy: Studying the use of structured light illumination for fluid inclusion analysis. [see publication list for more details].
- Dr. G. Wall, Dept. of Microbiology: Measuring protein size using FCS and fluorescence anisotropy.
- Dr. A.S. Klymchenko, Laboratoire de Pharmacologie et Physicochimie des interactions cellulaires et moleculaires, Faculte de Pharmacie, Universite Louis Pasteur, Illkirch, France: Use of ESIPT fluorophores for the measurement of polymer polarity and other physio-chemical parameters.
- Dr. Y. Rotchev, NCBES/School of Chemistry: development of fluorescence-based methods for the analysis of biomedical polymers, with emphasis of physiochemical measurements and drug elution profiles [see publication list for more details].
- Prof. John Parnell, Geofluids Research Group, Department of Geology and Petroleum Geology, University of Aberdeen: Fluorescence Lifetime Microscopy of hydrocarbon bearing fluid inclusions.
- Dr. H.-P. Nasheueur, Dept. of Biochemistry, NUIG: Fluorescence Correlation Spectroscopy (FCS) for protein-protein interactions [see publication list for more details].
- Dr. M. Feely, Dept. of Earth & Ocean Sciences, NUIG: Development of quantitative methods of analysis for hydrocarbon bearing fluid inclusions, Raman spectroscopy of aqueous fluid inclusions, gemstones, and mineral identification using spectroscopy. This collaboration has been ongoing since 1998 and is currently the focus of an RFP project [see publication list for more details].
- Dr. M. Madden, Dept. of Information Technology: Development of quantitative methods of analysis for illicit drugs and hazardous materials using Raman spectroscopy, chemometrics and machine learning. The current collaboration involves 2 Enterprise Ireland and one Marie-Curie grants. This collaboration has been ongoing since 2002 [see publication list for more details].
Past/completed Research Projects:
The following are some of the other research projects which we have been involved with in the past.
- Crude Oils & Fluid Inclusions: Rapid fluorescence lifetime analysis methods for characterizing crude petroleum oils. (More detail - petroleum fluorescence page). Fluorescence spectroscopy of hydrocarbon bearing microscopic fluid inclusions. We made the first fluorescence lifetime measurements from inclusions (July 2003) and have presented these preliminary results at the IMOG and MSI meetings in September 2003. This included the development of novel instrumentation and the use of chemometric analysis for petroleum composition. (More detail - fluid inclusion research page).
- Forensic applications of near-IR Raman spectroscopy: the use of multivariate analysis (Chemometrics) and Machine Learning (with M. Madden, IT Dept.) for identifying and predicting the concentrations of suspect materials. (More detail).
- Metal enhanced fluorescence (MEF) using nanopatterned substrates. This project started in August 2003 and was centred at the physics-chemistry-biology interface and aimed to develop new highly sensitive measurement tools for use in biology and medicine. Optical methods are ideal since they can be non-contact, non-destructive, and easily miniaturized. To deal with the ever-increasing need to measure smaller and smaller quantities of analytes, with very high accuracy we are going to combine nanotechnology with optical fluorescence. Fluorescence is the process by which certain molecules absorb light at one wavelength and then later re-emit light at another (usually longer) wavelength.
- Fluorescence Instrument Development: Development of time-resolved fluorescence instrumentation using Time Correlated Single Photon Counting (TCSPC) measurement methods. Development of portable phase-modulated fluorescence lifetime instrumentation for biomedical sensing applications. Development of a gated ICCD based FLIM system for widefield imaging applications.
- Development of lifetime based pH sensors for high accuracy pH measurement applications. ( More detail - pH sensing page)
- Other past small scale projects included:
- Analysis of Diamond Like Carbon (DLC) films with Enterprise Ireland.
- Analysis of Active Pharmaceutical Ingredients with Elan, Athlone.
- Analysis of chemical process samples, with Roche Ireland, Clarecastle.
- Study of colored inclusions extracted from MDMA tablets.
Tra Mor, Sheephaven Bay, Co. Donegal.

















